In this content, we learn about thermal runaway, the main cause of lithium-ion battery explosions, which happens when a battery starts generating heat faster than it can dissipate. We also learn about the Arrhenius equation, which explains how the rate of a reaction increases exponentially with temperature. Additionally, we discover that solutions to battery explosions involve pressure release valves, software battery controllers, different cathode materials, and separators that melt at higher temperatures. Sodium-ion batteries are also being tested, as they release far less oxygen when heated and tend to release their energy more slowly than lithium batteries. Finally, we learn that solving these problems does not require full knowledge of every single chemical reaction happening during thermal runaway, but rather a basic understanding of the problem and the creation of solutions based on that understanding.
Electric vehicles are not known for catching fire, but when they do, they can be extremely challenging to extinguish. Lithium-ion battery fires are not well understood, and scientists are still trying to determine the chemical processes that occur when these batteries catch fire.
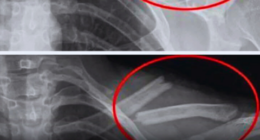
A nine-volt battery contains six individual cells, and an electric vehicle battery is made up of hundreds or thousands of cells. A battery fire starts in a single cell through a process called thermal runaway. X-ray footage shows gas building up inside the cell, which eventually ruptures the pressure release valve. Without a way to vent, the internal components of the battery continue to get hotter, and the steel outer casing stretches until it fails, resulting in an explosion.
These explosions are devastating and result in temperatures as high as a steel forge, melting everything in its path. One lithium-ion battery fire can require up to 20,000 gallons of water to extinguish.
The chemical reactions that occur during a lithium-ion battery fire are not well understood, which makes it difficult to prevent these fires from occurring or to effectively extinguish them when they do. Scientists are working hard to understand these reactions to make electric vehicles safer.
In conclusion, electric vehicle fires are rare but can be challenging to extinguish. Lithium-ion battery fires are not well understood, and scientists are still working to determine the chemical processes that occur during these fires. These fires can cause extensive damage and require large amounts of water to put out.
Lithium-ion battery fires are a rare but significant problem in electric vehicles. When one cell in the battery starts to thermally run away, it can heat surrounding cells, causing a chain reaction and resulting in a massive battery fire. Although there are groups of scientists and engineers trying to solve this problem, we still don’t fully understand the chemical processes that occur during a lithium battery fire.
The jelly roll is the structure of the battery, with the anode releasing electrons and the cathode absorbing them. These two layers are separated by an electrically insulating layer called the separator, which is key to understanding thermal runaway. The separator is made of plastic and is electrically insulating but chemically conductive, allowing lithium ions to pass through but not electrons.
When a cell in an EV starts to overheat and the temperature reaches about 130 degrees Celsius, the separator melts, causing the anode and cathode to make direct contact and creating a spark. This is called an internal short circuit or an ISC. Internal short circuits can be caused by various factors such as hammering a nail into the battery, a malfunctioning charger, overheating, or manufacturing defects.
Understanding the separator and the chemical processes that occur during a lithium-ion battery fire is essential to solving this problem. Although we have been working on this for a few decades, we still have a lot to learn. The key to preventing battery fires is to ensure that the battery doesn’t overheat, and this can be done by implementing a cooling system or using materials that can withstand high temperatures.
In conclusion, lithium-ion battery fires are a significant problem in electric vehicles, and they can spread quickly, making them challenging to extinguish. Internal short circuits can be caused by various factors, and understanding the separator and the chemical processes that occur during a lithium-ion battery fire is essential to solving this problem. Preventing battery fires can be achieved by implementing a cooling system or using materials that can withstand high temperatures.
Thermal runaway can cause a complete chemical cluster that generates a battery fire that’s difficult to put out. A battery that has experienced an internal short circuit (ISC) is prone to decompose at higher temperatures, releasing oxygen that enables combustion reactions. Unlike gasoline, batteries generate their oxygen when they burn, which is why they can keep reigniting unless the temperature is brought down.
The chemical reactions that occur during thermal runaway are complex, and even with decades of research, we still don’t fully understand them. This is partly due to the fast-changing battery technology, making it challenging to keep up with new battery materials and structures.
One way to engineer against thermal runaway is to understand the temperature behavior of the battery. A graph showing the temperature of a battery versus time shows that thermal runaway happens slowly and then all at once. The speed of the reaction is crucial to understanding the reactions, and this can be illustrated by mixing baking soda with room-temperature vinegar and vinegar at 57 degrees Celsius.
To prevent battery fires, we need to understand the chemical reactions that occur during thermal runaway. While this is a challenging task, we can engineer against thermal runaway by understanding the temperature behavior of the battery. Despite the challenges of keeping up with changing battery technology, we can use our understanding of the chemical reactions to design safer batteries.
Understanding the Role of Temperature in Reactions
Swedish chemist, Svante Arrhenius, in the 1800s, developed an equation to predict the rate at which a reaction would go faster at higher temperatures. This equation takes into account two important factors: K, the speed of the reaction, and T, the temperature in Kelvin raised to an exponent. In other words, as temperature increases, the speed of the reaction increases exponentially.
The Arrhenius Ouroboros
However, there is a positive feedback loop known as the Arrhenius ouroboros that can cause a reaction to spiral out of control. When a reaction gives off heat, the increase in temperature speeds up the reaction, which in turn gives off more heat, leading to a faster reaction and a subsequent increase in temperature. This positive feedback loop can drive temperatures in a battery from 200 to over a thousand degrees in just half a second, causing a battery fire.
Engineering Solutions to Thermal Runaway
Despite the challenges associated with understanding the underlying chemistry of thermal runaway, early scientists and engineers found ways to engineer solutions to these problems. For instance, a pressure-release valve can help release pressure in a battery to prevent explosions. Additionally, by engineering separators with a higher melting point, the occurrence of internal short circuits (ISC) can be reduced.
The Importance of Separators in Battery Safety
One of the most important components in battery safety is the separator, which is the electrically insulating layer that allows lithium ions to pass through but not electrons. When a cell overheats, the separator melts, which can cause the anode and cathode to make direct contact and spark an ISC. This can lead to a thermal runaway event, which is difficult to control.
Understanding the Conductivity of Materials
Understanding the conductivity of materials is also critical in preventing thermal runaway. Conductive materials like metals have lots of accessible empty orbitals that provide a path for electrons, whereas plastics don’t. Ions, on the other hand, can pass right through plastics without a path.
Conclusion
Although thermal runaway can be a complex and challenging problem to solve, understanding the underlying chemistry and engineering solutions can help prevent battery fires. With the rapid pace of battery technology advancements, it’s critical to stay on top of the latest developments and continue to improve safety measures.
Designing a battery with a high melting point separator is easier said than done, as changing the melting point without changing other properties requires a lot of trial and error. Nonetheless, progress has been made, and separators today melt at much higher temperatures than they used to. While pressure release valves and software battery controllers are the easiest, most obvious solutions to prevent thermal runaway, sometimes they don’t solve the problem all the time. In such cases, designing a whole new battery with a completely different ion, such as sodium, can be the solution. Sodium-ion batteries are being tested, and one cathode material that shows promise is sodium chromite, which releases far less oxygen when heated than similar cathodes in lithium-ion batteries. Sodium-ion batteries also tend to release their energy more slowly than lithium-ion batteries, providing more time for safety features to kick in and prevent explosions. While we don’t fully understand every single chemical reaction happening during thermal runaway, solutions such as pressure release valves, separators, and different cathode materials can be designed based on what we do understand. We can solve these problems before we fully understand them, just as solutions were engineered in the early days of lithium-ion batteries when scientists knew much less than they do now.
Don’t miss interesting posts on Famousbio